|
However, if energy must be added to an object in order to pull it apart, and if energy and mass are equivalent, then one could say that mass had to be added to pull it apart. It is, therefore, the amount of energy required to break the glue and pull the nucleus apart into its individual neutrons and protons. To understand this situation, one can think of the binding energy as the strength of the glue that holds the protons and neutrons together as a nucleus. The difference in mass, when expressed in energy (E) units according to German –American physicist Albert Einstein ’s (1879 –1955) formula E = mc 2 (where m refers to mass and c to speed of light), is called the binding energy of the nucleus. Second, when neutrons and protons are bound together as an atomic nucleus, the nucleus has less mass than the sum of the masses of the neutrons and protons. First, neutrons and protons do not happen to weigh exactly one amu apiece the proton actually weighs 1.0072765 amu and the neutron weighs 1.0086650 amu. (One amu is exactly one-twelfth of the mass of an atom of carbon-12, 12C, and is equal to approximately 1.66 ← × 10 -24 g.) There are two reasons why the mass number of an atom is different from its exact mass. It differs from the exact mass of the atom in atomic mass units, amu, which is often known and expressed to six decimal places. The mass number is always a whole number because it is a count of the particles. In the above example, isotope uranium-238 has 146 neutrons: 238 - 92.) 
(The difference between an atom ’s mass number and its atomic number provides the number of neutrons (n) that the atom possesses: n = A - Z. 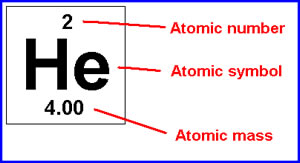
In the written symbol for a particular isotope, the mass number is written at the upper left of the symbol for the element, as in 238 92 U, where 92 is the atomic number (Z) of uranium (U) and 238 is the mass number (A) of this particular isotope. It is also called atomic mass number or nucleon number.ĭifferent isotopes of the same element have different mass numbers because their nuclei contain different numbers of neutrons. 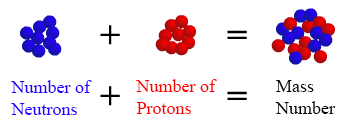
The mass number (A) of an atom, within physics and chemistry, is the total number of nucleons (protons and neutrons) in its nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
